The Choudhri Lab leads a translational research program at the intersection of neurosurgery, vascular biology, and biomedical engineering. Our work is driven by a single conviction: that the next generation of breakthroughs in brain disease will come from understanding the brain’s vascular and fluid systems with unprecedented precision—and then building the technologies to act on that understanding.
We pursue five interrelated research pillars, each designed to move from bench discovery to bedside impact. Our team includes neurosurgeons, neuroradiologists, biomedical engineers, and trainees from across disciplines, united by a commitment to rigorous science and clinical translation.

The brain’s blood vessels form one of the most intricate networks in the human body, yet many of the structures most relevant to neurovascular disease remain invisible to conventional imaging. Our lab develops and clinically validates next-generation imaging technologies—including Optical Coherence Tomography (OCT), high-resolution micro-DynaCT, and photon-counting CT—to visualize cerebrovascular anatomy at submillimeter resolution.
These tools allow us to see what was previously unseen: perforator arteries critical to surgical planning, venous microanatomy that governs drainage and pressure, and the subtle architectural changes that precede aneurysm rupture or fistula formation. By advancing the precision of diagnostic imaging, we aim to shift neurovascular care from pattern-based to precision-based, enabling surgeons to plan and execute treatments with a level of anatomical detail that fundamentally reduces risk and improves outcomes.
Clinical application: Our Micro-DynaCT intraoperative imaging protocol now enables real-time perforator vessel mapping during microsurgical aneurysm clipping—technology we have presented at the World Federation of Neurosurgery and the American Academy of Neurological Surgery.
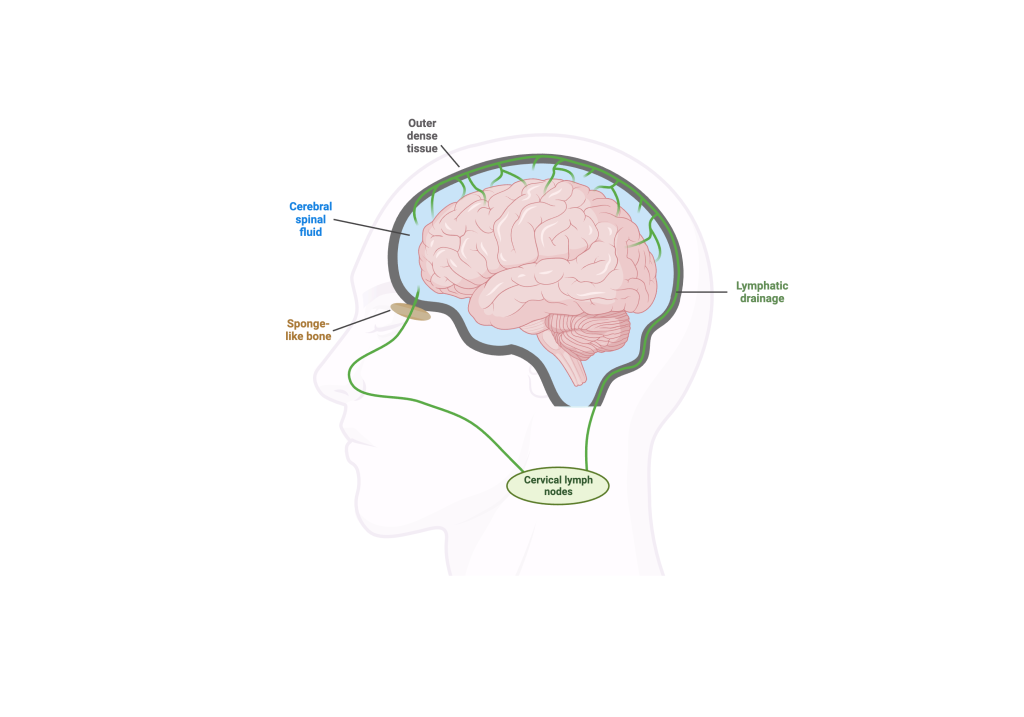
The brain’s glymphatic system—a recently discovered network of perivascular channels that clears metabolic waste through cerebrospinal fluid flow—represents one of the most exciting frontiers in neuroscience. Dysfunction of this system has been implicated in Alzheimer’s disease, normal pressure hydrocephalus, stroke recovery, and intracranial hypertension. Yet our ability to visualize, measure, and therapeutically modulate glymphatic function in living patients remains profoundly limited.
The Choudhri Lab is building a translational research program to bridge this gap. Our goals are threefold:
This research program is supported by planned R21 and K08 grant applications with the goal of establishing independently funded, R01-level investigation into glymphatic immunobiology.
Why it matters for donors: The glymphatic system sits at the intersection of neurodegenerative disease, cerebrovascular disease, and neuroimmunology—three of the highest-burden disease categories in medicine. Funding this work supports early-stage discovery with direct translational potential across multiple devastating conditions, including Alzheimer’s disease, which affects over 6 million Americans.
Cerebral venous disease—encompassing venous sinus stenosis, venous diverticula, dural arteriovenous fistulas, and idiopathic intracranial hypertension—remains underdiagnosed and undertreated. Many patients suffer for years with debilitating symptoms like pulsatile tinnitus, headaches, and vision loss before receiving an accurate diagnosis, in part because the venous system of the brain has historically received far less attention than the arterial system.
Our lab leverages one of the nation’s highest-volume cerebral venous referral practices to conduct clinical research aimed at defining diagnostic standards, optimizing treatment paradigms, and mapping the functional anatomy of the cerebral venous system with novel phlebography techniques. We have pioneered the use of balloon-assisted retrograde cerebral phlebography—a technique that provides unprecedented visualization of skull base venous structures—and published some of the earliest outcome data for endovascular treatment of venous sinus diverticula causing pulsatile tinnitus.
This research directly translates to patient care: every imaging innovation and treatment protocol developed in the lab is applied in the clinic, creating a tight feedback loop between discovery and outcomes.


Endovascular neurosurgery—the treatment of brain vascular disease through catheters navigated inside blood vessels—is one of the most technically demanding fields in medicine. Robotic platforms promise to transform this field by enhancing precision, reducing operator radiation exposure, enabling remote procedures, and democratizing access to expert care in underserved regions.
Our lab launched what became the nation’s largest longitudinal dataset for robotic cerebral angiography during our time at Penn, providing the foundational evidence base for robotic adoption in neurointerventional surgery. We continue to investigate the integration of robotic platforms with advanced imaging, haptic feedback, and artificial intelligence to build the next generation of semi-autonomous neurointerventional systems.
Vision: A future where a neurovascular emergency in a rural hospital can be treated by a specialist hundreds of miles away through a robotic interface—with the same precision as if they were in the room.
Brain-computer interfaces (BCIs) hold extraordinary promise for restoring function to patients with paralysis, locked-in syndrome, and neurodegenerative disease. Most current BCI approaches require open craniotomy to place electrodes directly on or in the brain—a barrier to widespread clinical adoption.
Our lab contributes to pioneering efforts in endovascular BCI—the concept of delivering recording and stimulation electrodes to the brain through the venous system using catheter-based techniques already familiar to neurointerventionalists. This work involves developing venous tractography methods to map the functional anatomy of the brain’s venous drainage, enabling the precise placement of endovascular electrodes near functionally important cortical regions without open surgery.
By combining our deep expertise in cerebral venous anatomy, advanced catheter navigation, and neurovascular imaging, the Choudhri Lab is uniquely positioned to contribute to this emerging field at the intersection of neurosurgery, neuroscience, and biomedical engineering.

The Choudhri Lab’s research program is fueled by the belief that today’s investments in basic and translational neurovascular science will yield tomorrow’s cures. From developing imaging technologies that reveal the brain’s hidden vascular architecture to pioneering robotic systems that could bring expert neurovascular care to every hospital in the country, our work sits at the edge of what is possible.
Philanthropic support accelerates every dimension of this mission—enabling us to recruit talented trainees, acquire cutting-edge equipment, launch pilot studies that attract federal grant funding, and bring new treatments from concept to clinical trial faster than traditional funding timelines allow.
If you share our vision for advancing the science and treatment of brain vascular disease, we invite you to learn more about how you can support this work.
NIH (PODCAST Trial, CHESS Trial, MPRESS Trial)
Semmes-Murphey Foundation (RAGE Trial)
McCabe Fund Award, University of Pennsylvania
Microvention Fellowship Grant
Cerebrovascular Research & Education Foundation (CREF)
Industry-Supported Clinical Trials: Cerus Endovascular, Galaxy Therapeutics, Sonorus Medical, Boston Scientific, J&J/Cerenovus
